
nanoCT of a healthy human prostate and bladder in the transverse plane performed in collaboration with Julia Bittencourt at the University of Florida in the Cohn lab
Overview
The main goals of the Strand lab are to create accurate cellular atlases of the human and mouse lower urinary tract, characterize the molecular and cellular alterations in human lower urinary tract disease, and build appropriate models of the human disease in novel mouse models.
Benign Prostatic Hyperplasia
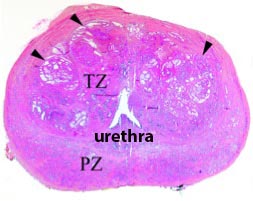
Cross-section of a prostate gland, showing the transition zone (TZ) and the peripheral zone (PZ)
A goal of the Strand laboratory is to understand the cellular and molecular biology of lower urinary tract symptoms (LUTS) due to benign prostatic hyperplasia (BPH) or bladder dysfunction. BPH/LUTS is present in the vast majority of men over 70 years old resulting in medical treatment or surgical intervention.
We focus on translational questions related to progression of BPH/LUTS in order to develop new targets for therapeutic intervention. In particular, we are interested in characterizing the cellular composition of particular phenotypes that are resistant to current therapies. We use a combination of single cell RNA sequencing, flow cytometry and immunohistochemistry on normal and diseased human specimens to build a cellular atlas of prostate and bladder disease.
Pathological Heterogeneity in BPH
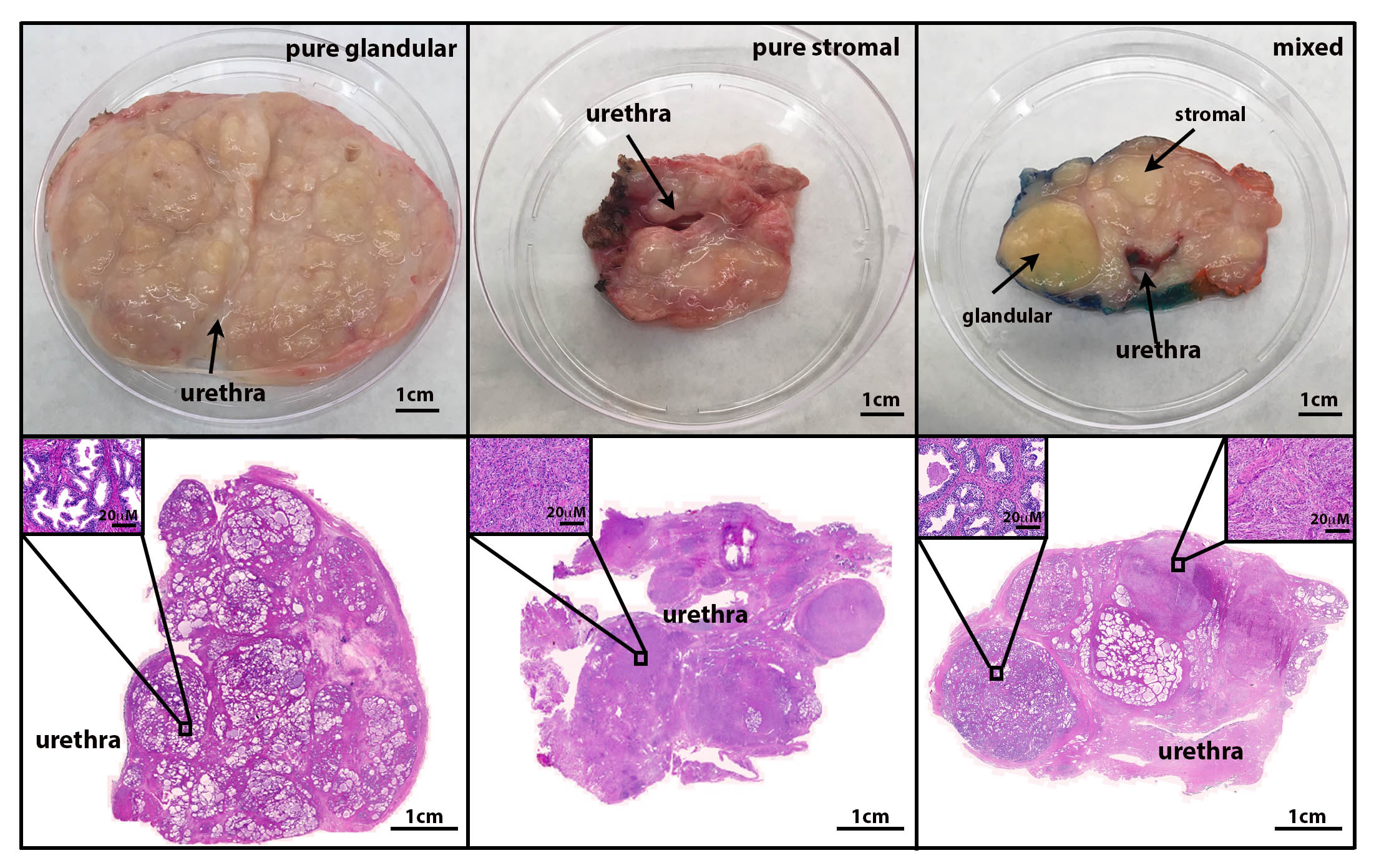
Phenotypic diversity in BPH. Left: glandular enlargement. Middle: Stromal enlargement. Right: Mixed type.
We believe that the basis for heterogeneous clinical responses in BPH is pathologic diversity. There are a number of phenotypes evident across and even within patients. In one area, a patient can display a stromal tumor while the other area display an epithelial nodule.
Our goal is to provide a rational basis for therapeutically treating these individual phenotypes by dissecting them macroscopically and then comparing their cell-specific molecular profiles to reduce the noise of cellular heterogeneity and inflammation. Our first goal is to identify a cell or cells of origin for the varying phenotypes by phenotyping the tissues with single cell RNA sequencing. This includes a comprehensive evaluation of the immune and inflammatory infiltrate as well as fluctuations in epithelial and stromal subpopulations.
We are subsequently isolating the cell types of interest and performing molecular profiling. Based on these targets, we will use our mouse and cell culture models to experimentally test our hypotheses.
Clinical Heterogeneity in BPH
It is clear from clinical trials of medications for BPH that personalized therapies will be needed due to the highly variable responses. Typically, patients complaining of lower urinary tract symptoms are placed on an alpha-adrenergic receptor blocker as a first line therapy due to its fast-acting relief of symptoms. These drugs relieve smooth muscle tone and are most effective in patients with early stage disease displaying a high smooth muscle to epithelial tissue composition. However, as the prostate enlarges, epithelial nodules predominate the tissue decreasing the likelihood of alpha-blocker efficacy.
The second-line therapy for BPH is a 5 alpha reductase inhibitor (5ARI), which induces epithelial apoptosis by decreasing the levels of intra-prostatic dihyrotestosterone (DHT). This therapy is effective at reducing prostate size by 25 percent in most patients.
Importantly, either therapy only reduces symptomatic progression by 34 percent, suggesting androgen-independent mechanisms of prostate growth and symptoms need to be targeted. Responses to current drugs are further reduced in patients with obesity, diabetes, and dyslipidemia.
These data suggest that there are further measures that are necessary for every patient, and completely novel measures necessary for individual patients. Our goal is to characterize the cellular composition of particular phenotypes, and more deeply understand the molecular changes driving these phenotypes. We use a combination of single cell RNA sequencing, flow cytometry, and immunohistochemistry on specimens from young organ donors and older men with prostatic enlargement.